|
1/15/2024 0 Comments 1920s reactivity trend
In the last stage of CP biogenesis, the prosegments are autocatalytically removed through nucleophilic attack by the active site residue Thr1 on the preceding peptide bond involving Gly(-1) 4, 5. Only three out of the seven different β subunits, namely β1, β2 and β5, bear N-terminal proteolytic active centres, and before CP maturation these are protected by propeptides 1, 2, 3. While the inactive α subunits build the two outer rings, the β subunits form the inner rings. Its seven different α and seven different β subunits assemble into four heptameric rings that are stacked on each other to form a hollow cylinder. The 20S proteasome core particle (CP) is the key non-lysosomal protease of eukaryotic cells. This work provides insights into the basic mechanism of proteolysis and propeptide autolysis, as well as the evolutionary pressures that drove the proteasome to become a threonine protease. Although a Thr1Ser mutant is active, it is less efficient compared with wild type because of the unfavourable orientation of Ser1 towards incoming substrates. Substitution of Thr1 by Cys disrupts the interaction with Lys33 and inactivates the proteasome. Here we use mutagenesis, X-ray crystallography and biochemical assays to suggest that Lys33 initiates nucleophilic attack of the propeptide by deprotonating the Thr1 hydroxyl group and that both residues together with Asp17 are part of a catalytic triad. However, the trigger for the self-activation and the reason for the strict conservation of threonine as the active site nucleophile remain enigmatic. 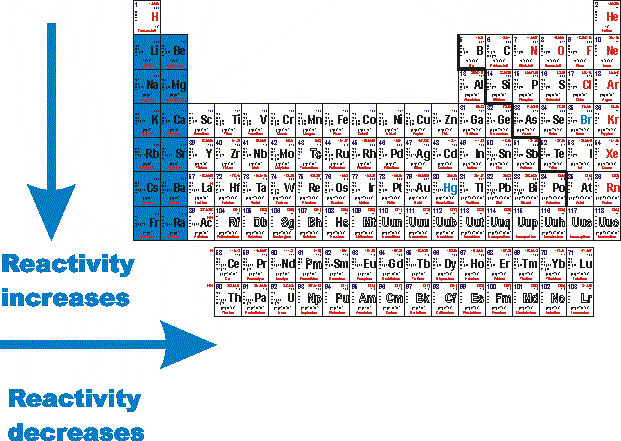 
The N-terminal propeptides protecting the active-site threonines are autocatalytically released only on completion of assembly. Biogenesis of the 20S proteasome is tightly regulated.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
